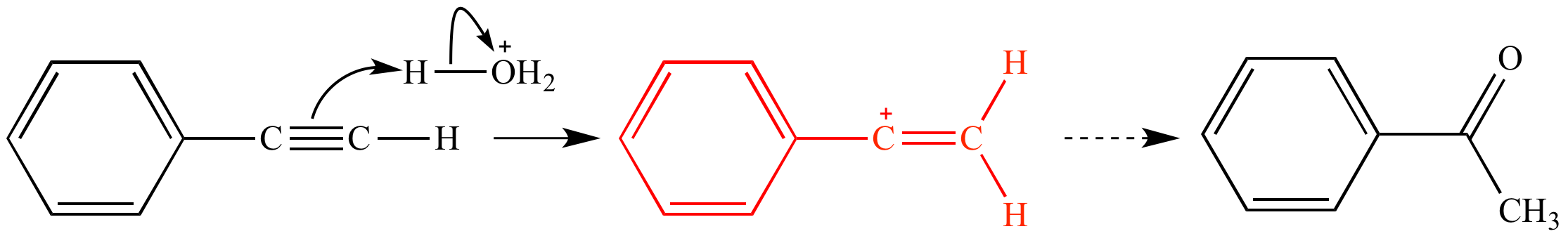
In the first mechanism step the alkyne is protonated by hydronium ion a strong acid to produce a resonance stabilized secondary vinylic carbocation shown in red.
A secondary vinylic carbocation is.
The general formula for vinyl group is r ch ch 2 in which both carbon atoms are bonded with double bond and r is attached at vinylic position.
Its empirical formula is c 2 h 3 more generally a vinylic cation is any disubstituted trivalent carbon where the carbon bearing the positive charge is part of a double bond and is sp hybridized in the chemical literature substituted vinylic cations are often referred to as vinyl cations and understood to.
The vinyl cation is a carbocation with the positive charge on an alkene carbon.
If in both resonance forms the formal charge of 1 is on a secondary carbon it also is a secondary allylic carbocation.
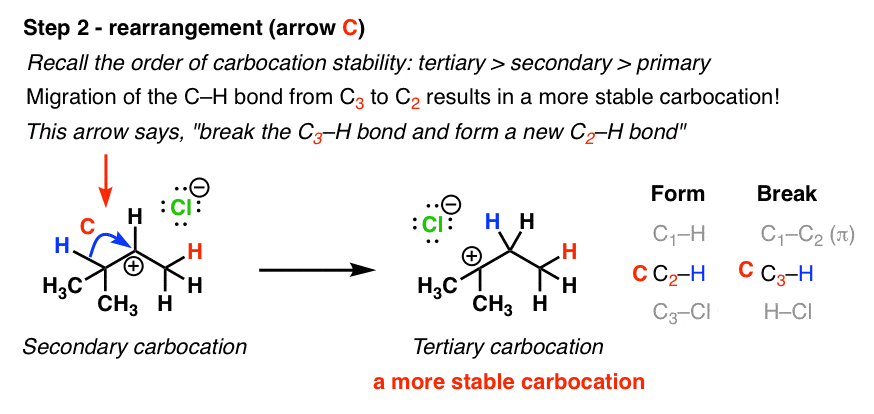
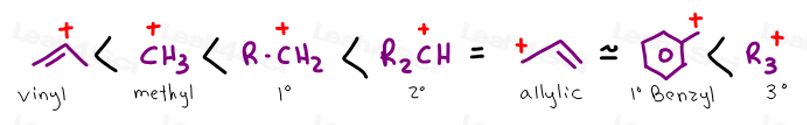
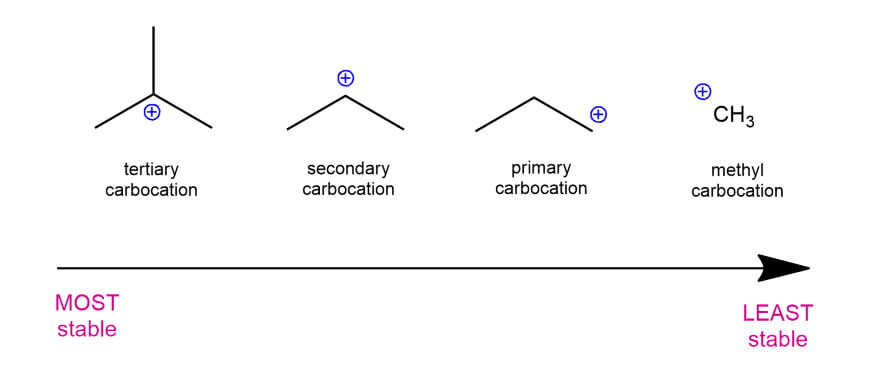
Secondary carbocations will require more energy than tertiary and primary carbocations will require the most energy.
If in the more stable of the two resonance forms of an allylic carbocation the formal charge of 1 is on a secondary carbon the allylic carbocation is called a secondary 2 allylic carbocation.
Since both carbon atoms form a double covalent bond so both are sp 2 hybridized.
The allylic position is also like a vinylic position.
The more stable the carbocation the lower the activation energy for reaching that intermediate will be.
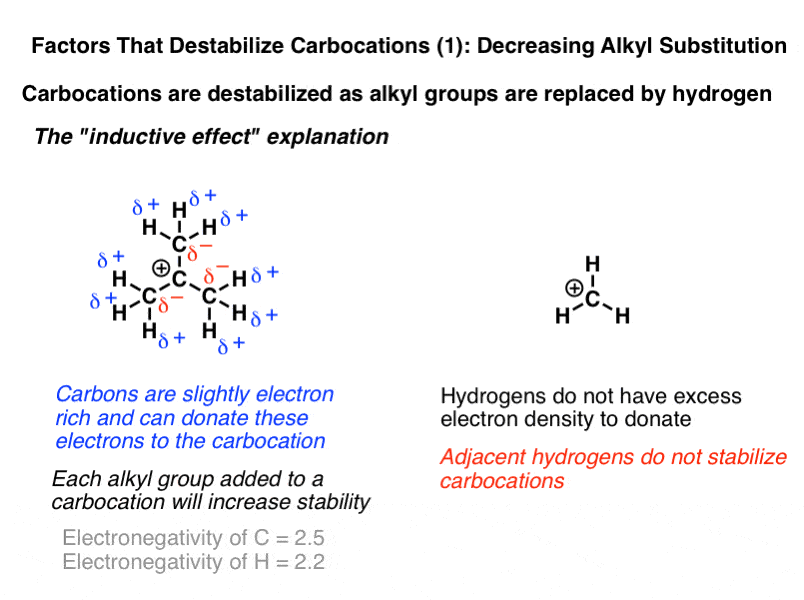
The more substituted a carbocation is the more stable it is.
We also acknowledge previous national science foundation support under grant numbers 1246120 1525057 and 1413739.
Acid catalyzed hydration of phenyl acetylene a terminal alkyne involves a vinylic carbocation intermediate.